Findings Time to Event
This report enables you to define events using one or more findings tests to be analyzed in a Time-to-Event analysis.
For events defined with more than one test code, it is assumed that these tests are scheduled on the same date/time. If a subject does not experience an event, they are censored on the date of their last available findings data.
Note: JMP Clinical uses a special protocol for data including non-unique Findings test names. Refer to How does JMP Clinical handle non-unique Findings test names? for more information.
Report Results Description
Running Findings Time to Event for Nicardipine generates the Report shown below. Refer to the Findings Time to Event requirements description for more information.
Note: In this example, VS was selected as the Findings Domain to Analyze, Hypertension was specified as the Event Name, and SYSBP >= 140 and DIABP >= 90 was entered as the Event Definition.

The following elements are generated in this report:
Hypertension:
| • | The name of this report corresponds to the Event Name supplied in the dialog. If this is not supplied, it is called Domain Event where Domain is determined by the Findings Domain to Analyze selected in the dialog. For example, if the event is defined using the VS domain, the section is called VS Event. This section contains a Kaplan-Meier analysis of the time to the defined event. |
This analysis compares the time until the event among treatments in the study. Subjects not experiencing the defined event are censored at the date of their last available findings data.
See Survival Plot for more information.
General
| • | Click  to view the associated data tables. Refer to View Data for more information. to view the associated data tables. Refer to View Data for more information. |
| • | Click  to generate a standardized pdf- or rtf-formatted report containing the plots and charts of selected sections. to generate a standardized pdf- or rtf-formatted report containing the plots and charts of selected sections. |
| • | Click  to generate a JMP Live report. Refer to Create Live Report for more information. to generate a JMP Live report. Refer to Create Live Report for more information. |
| • | Click  to take notes, and store them in a central location. Refer to Add Notes for more information. to take notes, and store them in a central location. Refer to Add Notes for more information. |
| • | Click  to read user-generated notes. Refer to View Notes for more information. to read user-generated notes. Refer to View Notes for more information. |
| • | Click  to open and view the Subject Explorer/Review Subject Filter. to open and view the Subject Explorer/Review Subject Filter. |
| • | Click  to specify Derived Population Flags that enable you to divided the subject population into two distinct groups based on whether they meet very specific criteria. to specify Derived Population Flags that enable you to divided the subject population into two distinct groups based on whether they meet very specific criteria. |
| • | Click the arrow to reopen the completed report dialog used to generate this output. |
| • | Click the gray border to the left of the Options tab to open a dynamic report navigator that lists all of the reports in the review. Refer to Report Navigator for more information. |
Report Options
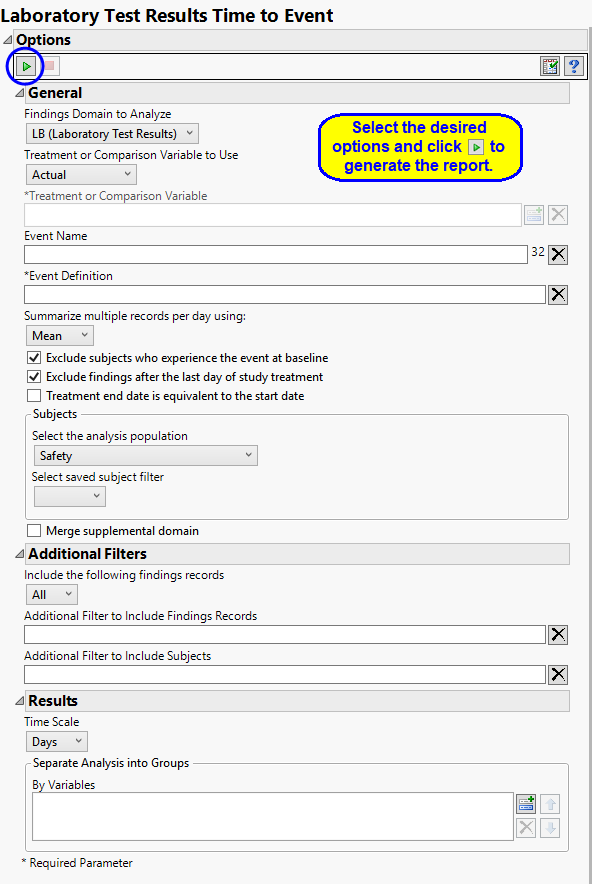
Findings Domains
Use the Findings Domain to Analyze option to specify whether to plot the distribution of measurements from either the Electrocardiogram (EG), Laboratory (LB), or Vital Signs (VS) findings domains. LB is selected by default.
Treatment or Comparison Variable:
The primary goal of clinical trials is to distinguish treatment effects when reporting and analyzing trial results. Treatments are defined by specific values in the treatment or comparison variables of the CDISC models. These variables are specified in this report using the Treatment or Comparison Variable to Use andTreatment or Comparison Variable options.
Available variables include Planned, which is selected when the treatments patients received exactly match what was planned and Actual, which is selected when treatment deviates from what was planned.
You can also specify a variable other than the ARM or TRTxxP (planned treatment) or ACTARM or TRTxxA (actual treatment) from the CDISC models as a surrogate variable to serve as a comparator. Finally you can select None to plot the data without segregating it by a treatment variable.
See Treatment or Comparison Variable to Use, Treatment or Comparison Variable for more information.
Events
Use the following options to name and define the event to be analyzed. You must also specify the conditions under which events are to be considered and reported.
You must name enter a name and define the boundaries by which the event is defined. For example, if elevated blood pressure is an expected event, you might enter Hypertension in the Event Name text box and then define the parameters to be used to define hypertension (systolic blood pressure is greater than 140 and the diastolic pressure is greater than 90, for example). Refer to Event Definition for more information about defining an event.
JMP Clinical summarizes events occurring multiple times on the same day rather than individually reporting each occurrence. The Summarize multiple records per day using: option enables you to specify the statistic used to summarize the records for each day.
You can restrict the analysis to those subjects who first experience the event only during the course of the study. If the Hypertension is the event being reported, you might want to eliminate subjects who normally have an elevated blood pressure. Selecting the Exclude subjects who experience the event at baseline option excludes all subjects entering the study already meeting the specified criteria for Hypertension. Alternatively, you might also want to exclude subjects who first experience the event only after the study has concluded. Selecting the Exclude findings after the last day of study treatment option excludes those subjects.
Check the Treatment end date is equivalent to the start date if the treatment end date (EXTENDTC) is missing from the data. In this case, it is assumed that all treatments were given on the same day and that the treatment start date can be used instead.
Filtering the Data:
Filters enable you to restrict the analysis to a specific subset of subjects and/or findings records, based on values within variables. You can also filter based on population flags (Safety is selected by default) within the study data.
If there is a supplemental domain (SUPPXX) associated with your study, you can opt to merge the non-standard data contained therein into your data.
See Select the analysis population, Select saved subject filter1, Merge supplemental domain, Include the following findings records:, Additional Filter to Include Findings Records, and Additional Filter to Include Subjects2 or more information.
Results
By default, time is measured in days. However, you can change the Time Scale to measure time in weeks. This option is useful for assessing report graphics for exceptionally long studies.
You can also subdivide the subjects and run analyses for distinct groups by specifying one or more By Variables.